ABOUT
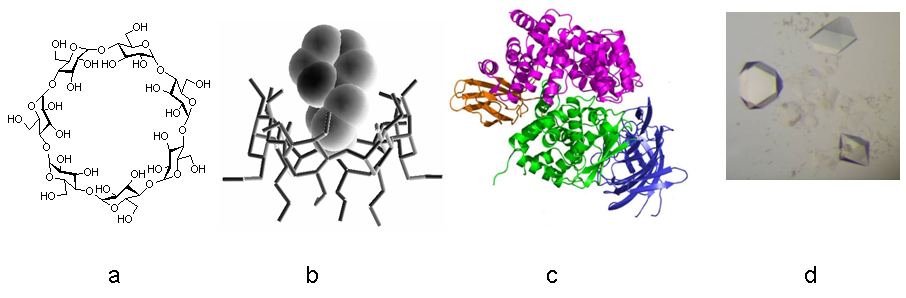
The laboratory focuses on supramolecular systems comprising chemically modified cyclodextrin (CD) host macrocycles (hollow oligomeric carbohydrates) and various bioactive or photoactive guest molecules, as well as on the structure of biological macromolecules. The aim is to unravel the structural elements that dictate key properties of such systems using NMR spectroscopy in solution and X-ray crystallography in the solid state. The laboratory also has strong expertise in cyclodextrin chemistry and in macromolecular crystallization.
Main objectives are the a) determination of structure and intermolecular interactions of supramolecular systems, b) development of CD-based multimodal drug transport and release systems and the study of their photophysical and biological properties, c) development of macromolecular crystallization methods and d) crystal structure analysis of medically important biological macromolecules and their complexes.
Main Scientific Directions:
- Cyclodextrin (CD) chemistry: the group specializes in regioselective chemical modifications of natural CDs to afford derivatives of higher aqueous solubility, longer host cavity, increased guest specificity and self-assembly properties to nanoparticles. The derivatives are intended for numerous nano bio applications such as cell membrane crossing, drug transport, antibacterial activity, metal binding, photo/chemotoxic effects.
- Structure and interactions in solution and crystalline state: the final properties of the designed CD hosts in complex with selected guest molecules, depend on the structure and the developed interactions. Therefore, we focus on issues such as supramolecular assembly in the solid state and in solution, complexation modes and stoichiometries, guest recognition, enantioselectivity/enantiospecificity, inclusion binding equilibria and release studies. Moreover, for photoactive guest molecules, we investigate how encapsulation in CDs controls and regulates their photophysical properties in aqueous solution, such as absorption, fluorescence, photostability, intracellular localization and effect on cell lines under irradiation, and photoswitching behavior.
- Structural analysis of biological macromolecules: Knowledge of the structure of biological macromolecules can explain their function, reveal mechanisms and pathways in the cells and be eventually applied, e.g. to improve the design of drugs. Structures solved include several 2[4Fe4S] ferredoxins from pathogenic bacteria, the Endoplasmic Reticulum aminopeptidase ERAP2 and the insulin-regulated aminopeptidase IRAP that participate in the regulation of the immune response, and the corresponding structures with rationally designed inhibitors.
- New crystallization methodology for biological macromolecules: Crystallization is a complex, multi-parametric process which is poorly understood and controlled. The group is developing novel methodology as well as striving for a better understanding of macromolecular crystallogenesis, including research into substances and materials promoting the heterogeneous nucleation of crystals, and the use of phase diagrams, Genetic Algorithms and calorimetry for optimization and prediction of crystallization conditions.
Moreover, the laboratory is also committed to education and training of graduate students and post-doctoral fellows in interdisciplinary research ranging from organic synthesis to structure determination by X-ray crystallography in the crystalline state and by NMR spectroscopy in solution.